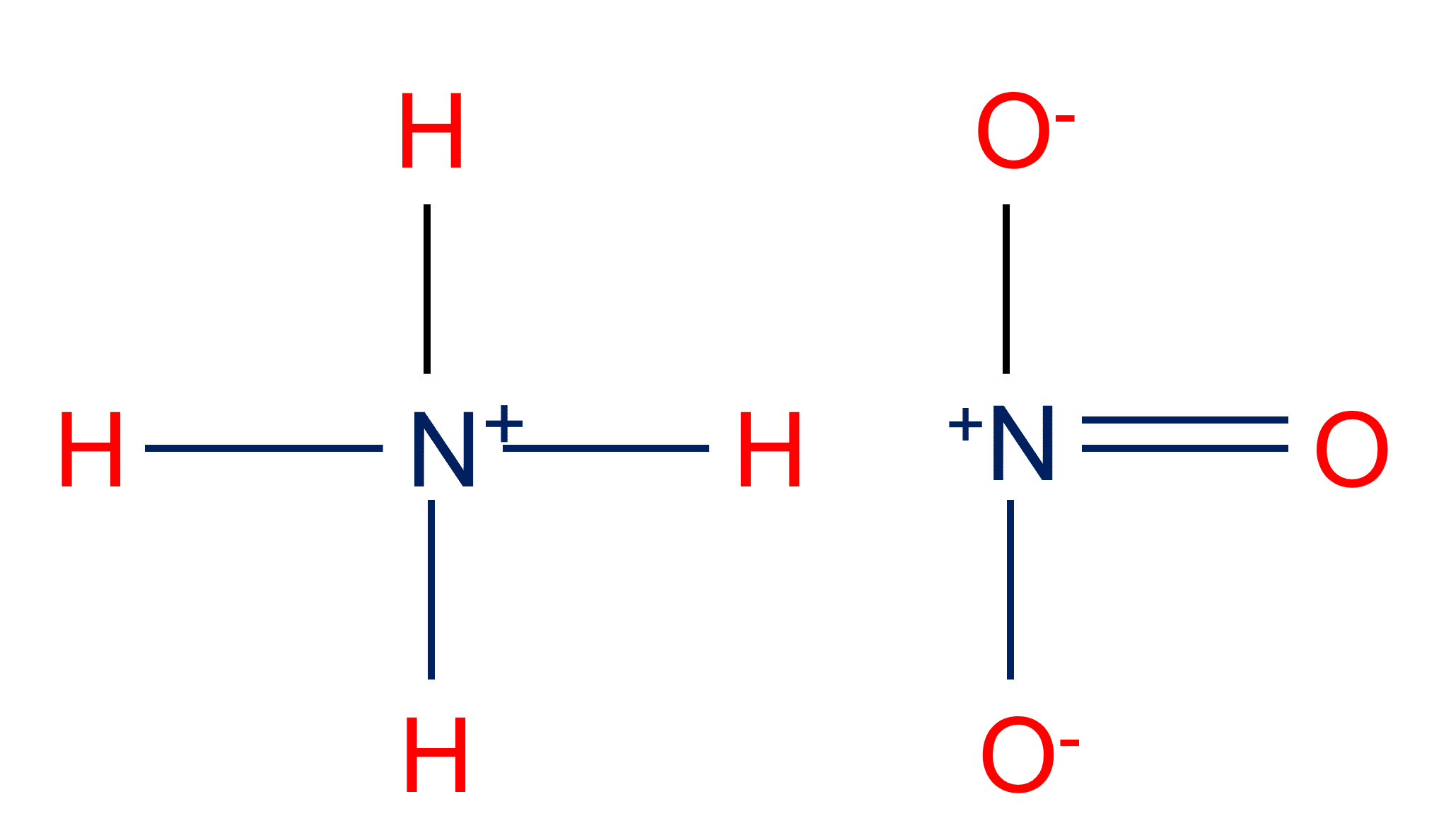
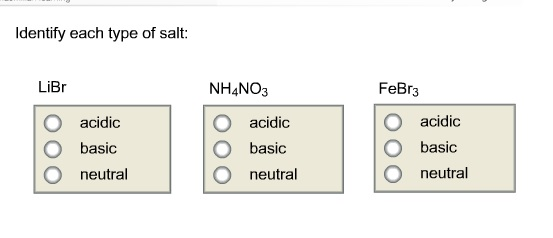
Predict if the solutions of the following salts are neutral, acidic or basic. NaCl, KBr, NaCN, NH4NO3,NaNO2 and KF

Predict if the solutions of the following salts are neutral, acidic or basic. NaCl, KBr, NaCN, NH4NO3,NaNO2 and KF

Ammonium Nitrate (NH<sub>4</sub>NO<sub>3</sub>) - Structure, Preparation, Physical and Chemical Properties, Uses with FAQs of Ammonium Nitrate

SOLVED: CN- NH4CL NaNO3, NH4+ Na + NaOH; HCN, NH4NO3, H2C03, NH3; CI -, HSOA HCIO3 Fill in the blanks below. Some answers from the list may be used more than once.

SOLVED: Write an ionic equations for the hydrolysis reactions of NH4NO3 and Na2SO3. Predict the pH of the solution equal 7, greater than 7, less than7).

OneClass: Ammonium nitrate, NH4NO3, is a salt formed from the neutralization of the weak base ammonia...

Uses of ammonia nitric acid ammonium nitrate salts fertilisers preparation pollution eutrophication gcse igcse KS4 science chemistry O level revision notes revising

Ammonium nitrate, NH4NO3, is a salt formed from the neutralization of the weak base ammonia with the strong acid nitric acid. Given that the value of Kb for ammonia is 1.8 x