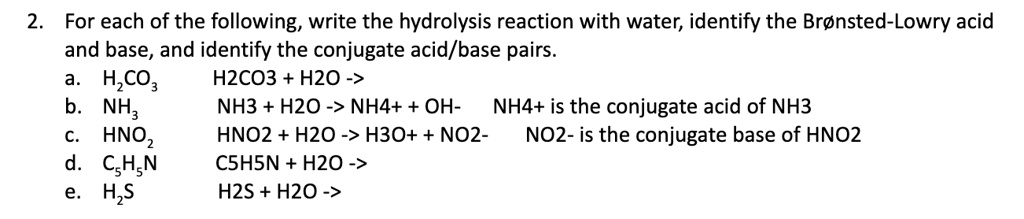
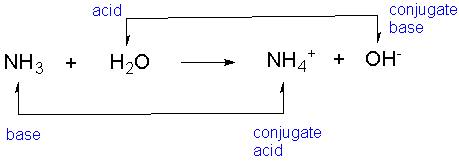
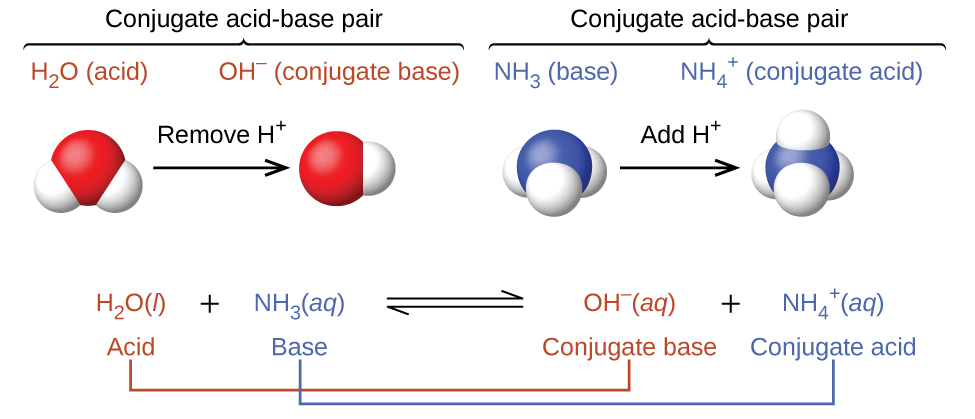
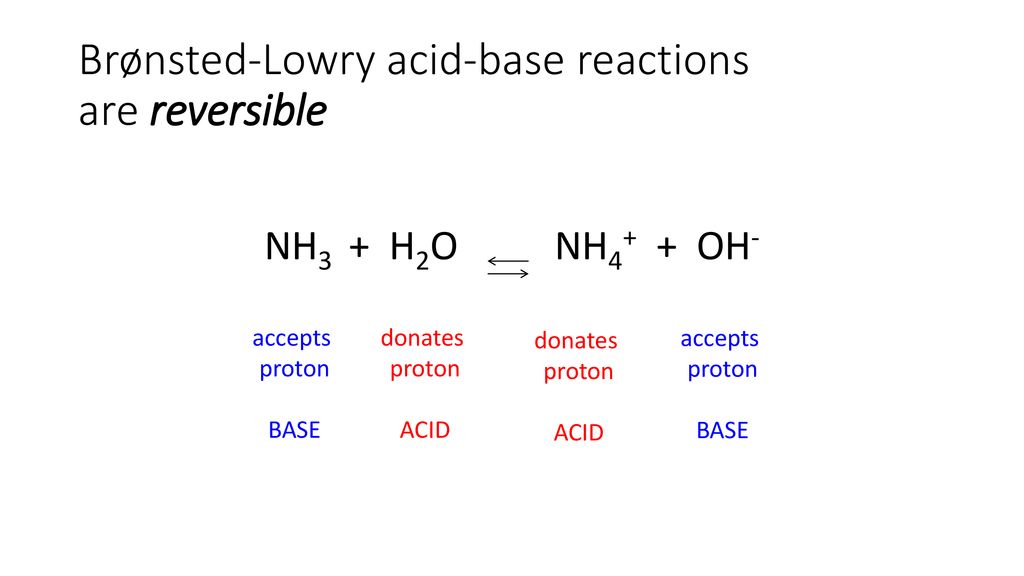
SOLVED: In the reaction NH3 (aq) + H2O (I) <–> NH4+ (aq) + OH- (aq), which is the conjugate acid-base pair? OH- NH3 NHA+, OH- NH3, H2O NH4+, NH3
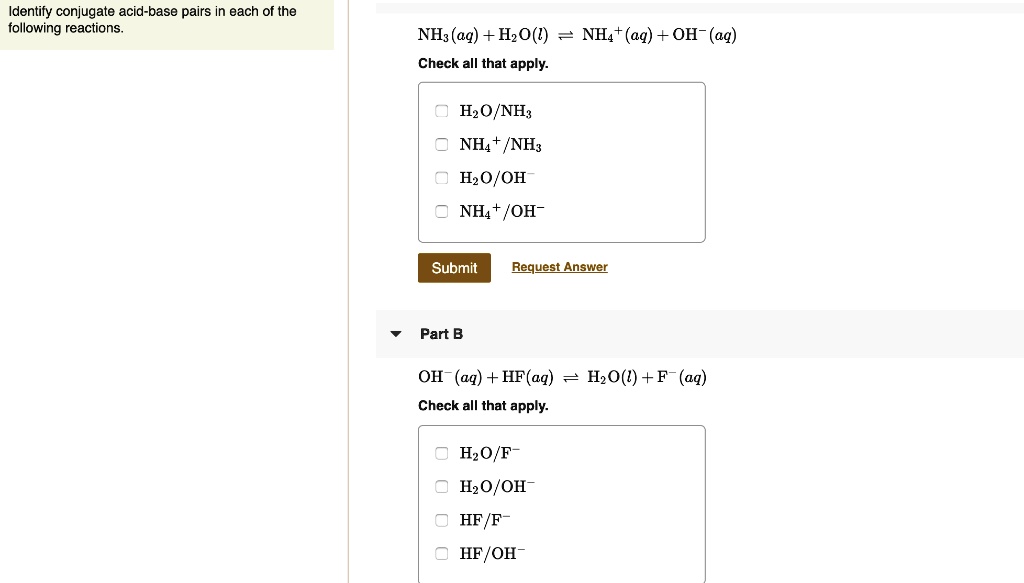
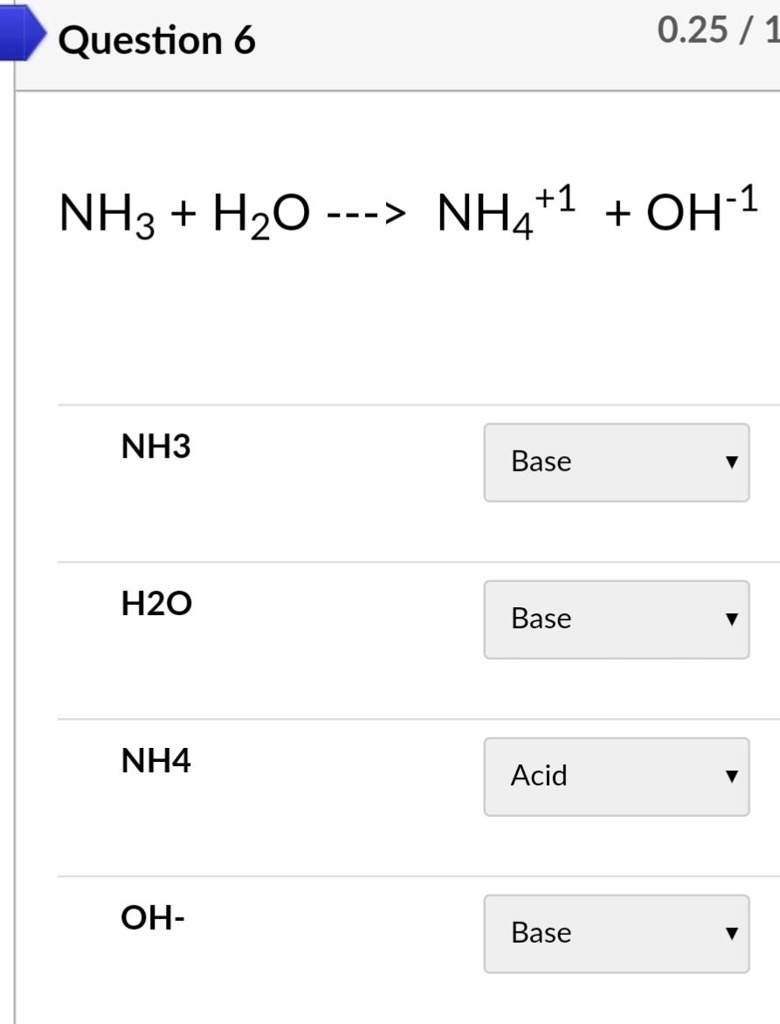
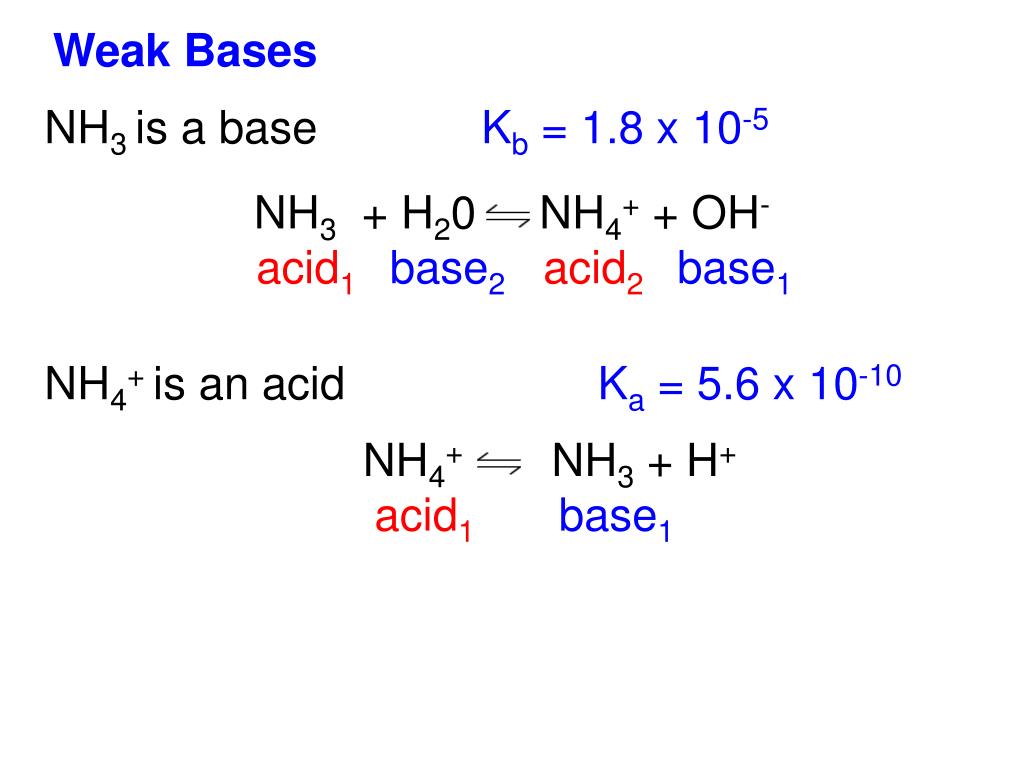
SOLVED: Identify conjugate acid-base pairs in each of the following reactions NH; (aq) + HzO() Check all that apply: NH4 + (aq) + OH- (aq) HzO/NH: NH4 NHz HzO/OH NH4 + /OH-
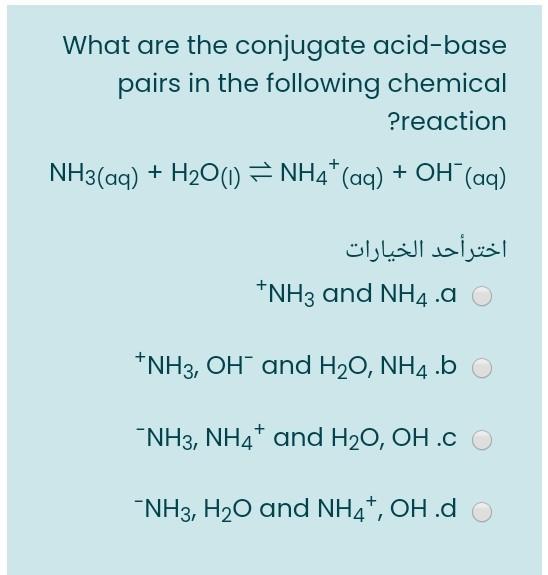
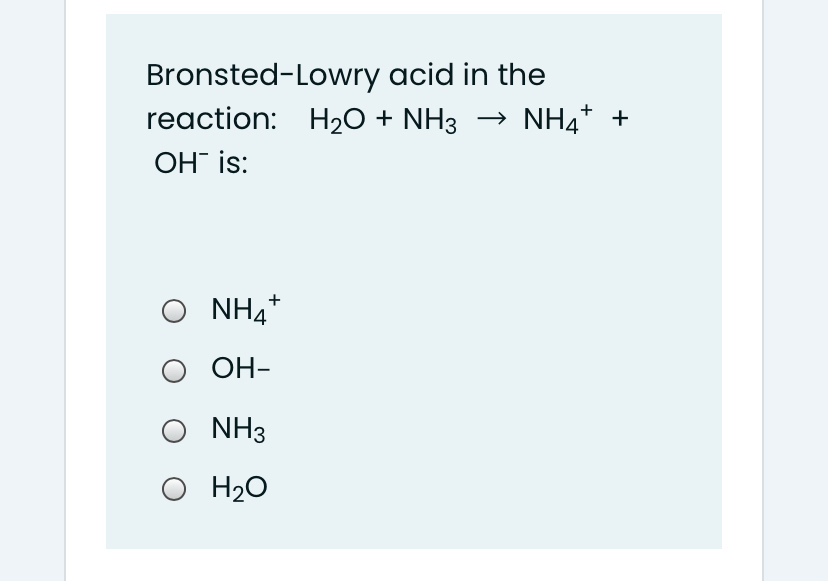
Solved] Consider the reaction below: NH3 + H20 --> NH4* + OH Who is the conjugate acid in the forward reaction? | Course Hero